Key Takeaways
- MedTech software development sits at the intersection of strict regulation, patient safety, and high UX expectations.
- The SaMD market is growing rapidly, heavily driven by AI/ML-based solutions and increasing FDA approvals.
- Compliance by design (FDA, ISO 13485, ISO 14971, IEC 62304, HIPAA) is non‑negotiable and must be embedded from the first sprint.
- Agile delivery, cross‑functional teams, rigorous testing, and living documentation are the four practices that most strongly correlate with successful MedTech projects.
- User-Centered Design and usability are critical - in healthcare, bad UX can directly translate into clinical risk and safety incidents.
Creating medical software is one of the most demanding areas of engineering - it combines strict regulatory requirements with the need to deliver an excellent user experience. In this guide, we walk through the key MedTech software categories, compliance standards, and the most important practices that drive successful projects.
Key Categories of MedTech Software
Before we dive into best practices, it’s important to be clear about the types of software we’re talking about. The MedTech software ecosystem can be split into several main categories, each with different regulatory and technical requirements.
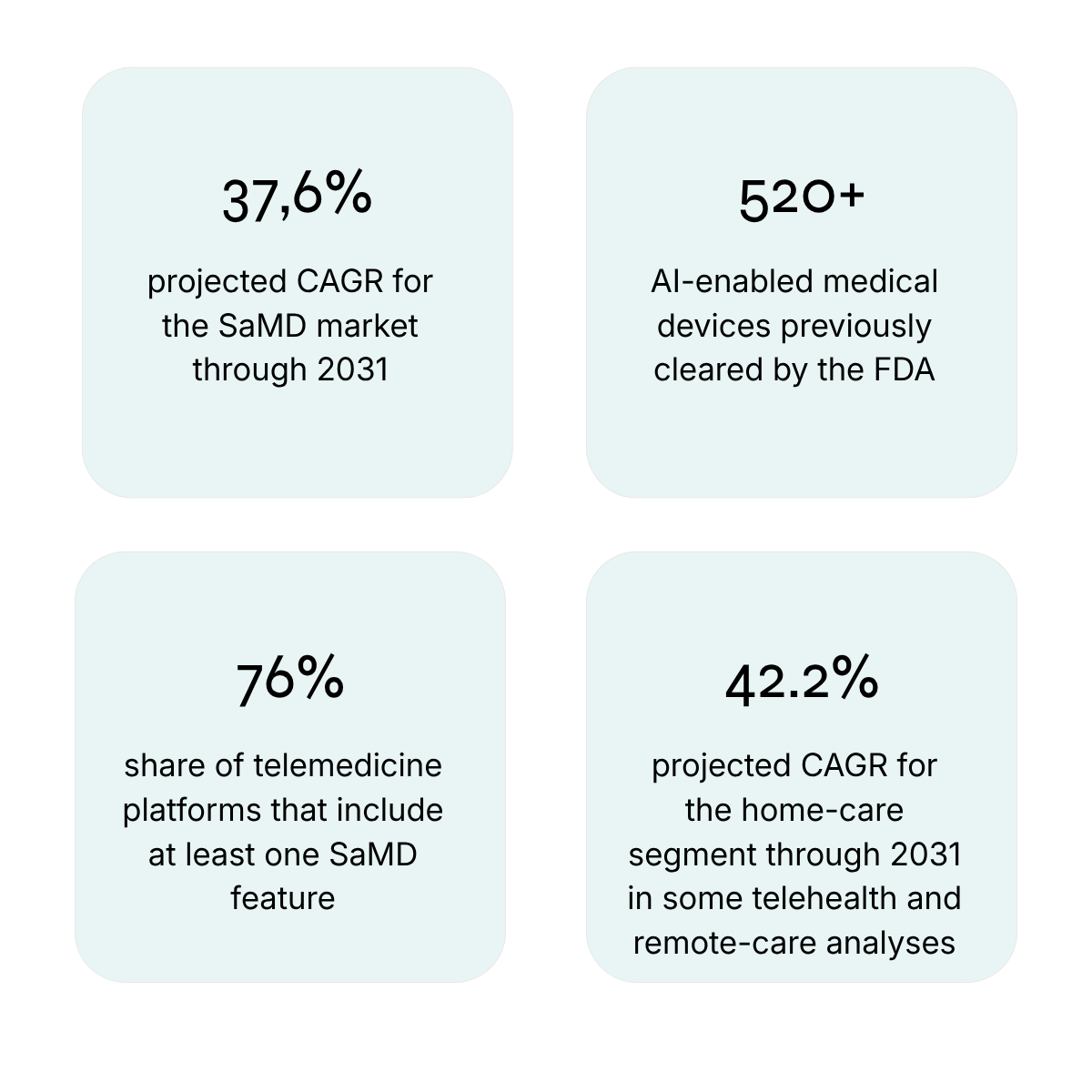
Software as a Medical Device (SaMD)
These are applications that independently perform medical functions - such as diagnostics, patient monitoring, or clinical decision support. The SaMD market is growing at around 37.62% CAGR and is strongly driven by AI and machine learning innovations. The FDA has now authorized hundreds of AI/ML-enabled medical devices (the latest lists show around 950 devices in total, up from just over 500 a few years ago), which underscores the strategic role of this category.
Electronic Health Records (EHR)
EHR systems support patient data management and care coordination across hospitals and clinics. They account for a significant share of digital health spending, and large health systems consistently show that well-implemented EHR programs can improve guideline adherence and care quality. If you’d like to explore this area further, check out our article on healthcare integration software (mentioned in the original Polish text).
Telemedicine Platforms
The COVID-19 pandemic dramatically accelerated the adoption of telemedicine, and today a large majority of telehealth platforms include at least one SaMD-like function, such as remote diagnostics or remote patient monitoring. The home-care segment is projected by some market studies to grow at more than 40% CAGR over the coming years, making it one of the fastest-growing areas of the virtual care market.
Clinical Decision Support Systems (CDSS)
CDSS tools help clinicians make decisions based on clinical data and patient-specific information. When combined with AI, CDSS is transforming how clinicians approach diagnosis and treatment planning, especially in fields like radiology, cardiology, and neurology where most AI-enabled devices are currently used.
Mobile Health Applications
Mobile health apps support health tracking, chronic disease management, and wellness. They turn patients into active participants in their own care, which is especially important in value-based care models where outcomes and engagement are key.
Hospital Information Systems (HIS) and Practice Management Software
Hospital Information Systems (HIS) and practice management platforms sit at the core of operational workflows in hospitals and clinics. They handle scheduling, admissions and discharge, bed management, billing, and clinical documentation.
For MedTech vendors, integration with existing HIS or practice management stacks is often a hard requirement in RFPs, because it directly affects clinician workflows and data visibility.
Medical Imaging and Diagnostic Systems (PACS/RIS and AI Imaging)
Medical imaging software - including Picture Archiving and Communication Systems (PACS) and Radiology Information Systems (RIS) - manages medical images and radiology workflows across the enterprise.
AI‑powered diagnostic tools frequently plug into PACS/RIS as SaMD modules, for example in radiology, cardiology, and oncology, where most FDA‑cleared AI/ML devices currently sit.
Remote Patient Monitoring (RPM) and Connected Devices
Remote Patient Monitoring (RPM) platforms collect real‑time data from connected medical devices and wearables, feeding it into dashboards for clinicians and sometimes directly into EHR/CDSS.
RPM is a key engine behind the growth of home-care and virtual wards, and often combines hardware, mobile apps, and cloud backends into a single regulated product ecosystem.
Regulatory Compliance: Standards and Requirements
In MedTech, compliance isn’t optional - it’s the foundation of every project. Failing to meet regulatory expectations can lead to product withdrawals, financial penalties, and most importantly, patient safety risks. Below are the core regulations and standards you need to know:
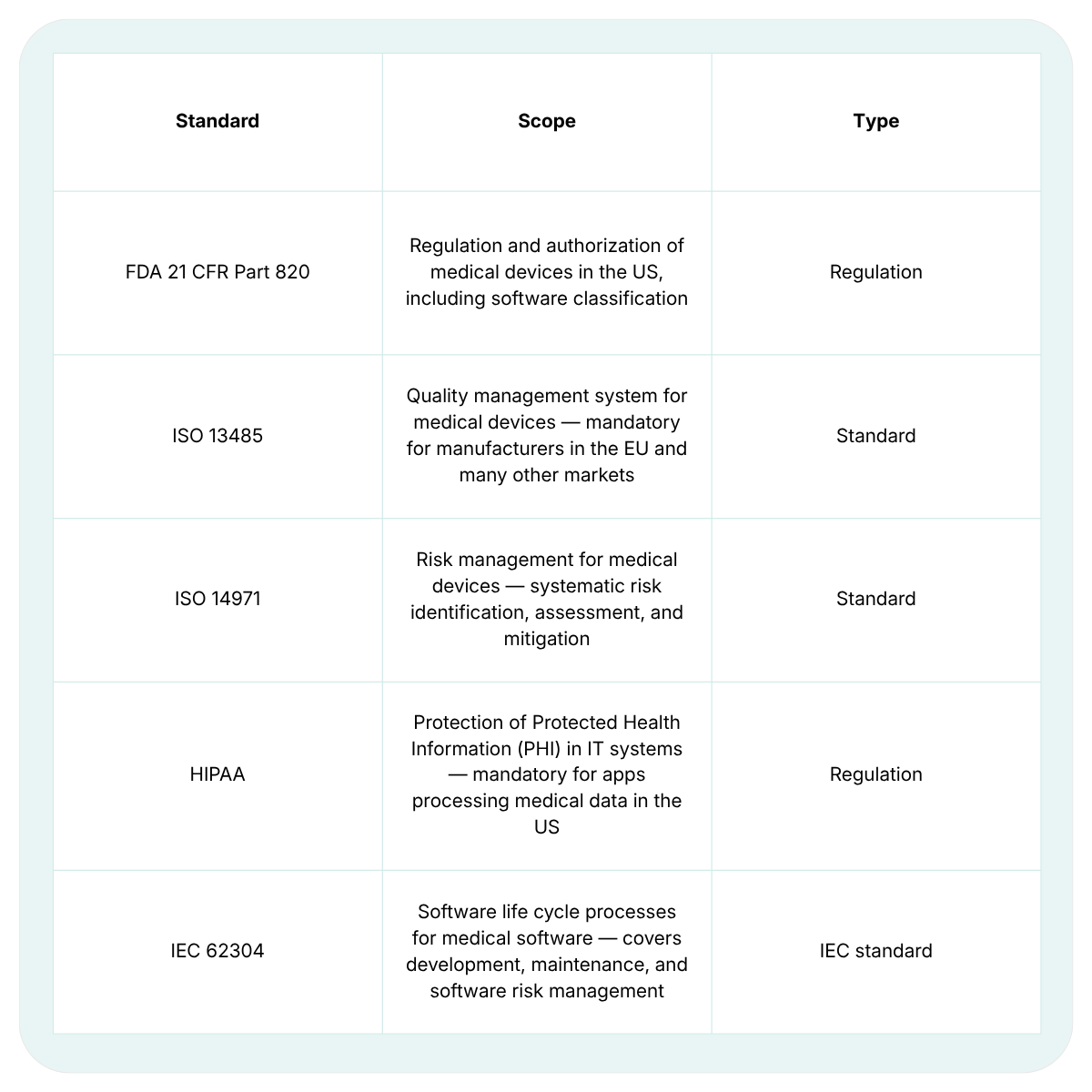
The key takeaway: compliance has to be built into the development process from the first sprint. Trying to bolt it on at the end is far more expensive and significantly riskier. For a deeper dive into how this works in practice, see our article on Agile practices in the software development lifecycle referenced in the original text.
Four Best Practices in the Development Process
Based on what we’ve seen across successful and failed MedTech projects, four practices have the biggest impact on the final outcome.
Agile Methodology with Compliance in the Loop
Organizations that use Agile methodologies report average project success rates in the mid‑70% range, which aligns with multiple industry surveys. In MedTech, Agile means iterative progress, continuous feedback collection, and fast adaptation to changing product and regulatory requirements. Every sprint should include a compliance checkpoint, not just a functional review. For a practical guide, see our resource on navigating compliance in MedTech digital products.
Cross-Functional Teams: Developers, Regulatory Experts, Clinicians
Having a cross-functional team isn’t a “nice to have” but it’s a requirement. Developers understand the technology, regulatory experts know FDA, ISO, and local requirements, and clinicians bring real-world knowledge of user needs and workflows. Real-world experience shows that cross-functional collaboration leads to better decision quality and stronger adherence to standards.
Rigorous Testing at Every Stage
Testing protocols in MedTech should cover at least:
- Unit testing – verifying component logic
- Integration testing – ensuring system interoperability
- Performance testing – checking stability and behavior under load
Testing isn’t the final step and integral part of every phase of the medtech software life cycle.
Documentation as a Strategic Asset
Thorough documentation supports compliance, enables future updates, and is essential during audits. Treat documentation as a living project artifact - kept up to date throughout the entire product lifecycle, not written only at the end.
User Experience and Usability in MedTech
Poor UX is estimated to cost enterprises around $1.4 trillion globally each year, and in healthcare the consequences are even more serious. Interface-related errors can directly affect patient safety. Here are the five pillars of good UX in medical software:
- Interface simplicity – minimize cognitive load for users working under time pressure in clinical environments.
- Accessibility – compliance with ADA, support for screen readers, and keyboard navigation. Good accessibility broadens your user base and improves adoption metrics.
- Feedback mechanisms – instant confirmations, alerts, and visual cues are critical where timing and clarity of information directly affect medical decisions.
- Training and support – comprehensive training materials, FAQs, and responsive support reduce frustration and increase adoption rates in healthcare organizations.
- Iterative usability testing – involve real users (doctors, nurses, patients) at every stage to catch issues early and validate solutions.
User-Centered Design isn’t just a project phase but a philosophy that runs through the entire process, from discovery through post-launch iterations.
Summary
Success in MedTech software development comes down to combining four elements:
1. A deep understanding of medical software categories and their contexts.
2. Embedded compliance from day one, instead of a last-minute retrofit.
3. Agile delivery supported by cross-functional teams that combine engineering, regulatory, and clinical expertise.
4. User-centered UX designed around the needs of real end users, backed by accessibility, training, and continuous usability testing.
Investing in a high-quality process today leads to better patient outcomes and lower regulatory risk tomorrow.
Turn your MedTech roadmap into a compliant delivery plan
If you’re planning a new healthcare product and want to stress‑test your architecture and compliance approach, let’s talk.
In a 45‑minute discovery call we’ll walk through your current roadmap, identify the riskiest assumptions, and outline a practical next step for your team. Book a call




.svg)










.png)



















